WHEREABOUTS/
Conference season is about to begin, and Cost Curve is hitting the road. I enjoy meeting newsletter readers in person, given that you’re a thoughtful group.
So if it looks like we’ll be in the same place at the same time, please let me know. Coffee is on me.
Here’s the lineup over the next six weeks or so.
Feb. 17: PhRMA Forum in DC
Feb. 23 and 24: AAM Access! I’ll be doing a fireside chat on Tuesday.
March 9 and 10. PCMA Business Forum. Speaking time TBD.
March 16 to 18. Drug Channels Leadership Forum. I’ll moderate a session on Tuesday morning.
March 26. MassBio State of Possible Conference. Look for me on an afternoon panel.
Things slow down a little in April, but only a little. I’m on the program at the Tuft CEVR annual meeting and the COA Community Oncology Conference. More on those events as they get closer.
This Week in Cost Curve Apex:
Pfizer Cut the Price of Certain Forms of Its Blockbuster Breast Cancer Med Ibrance. Here’s One Possible Reason Why.
IQVIA Offers a Sneak Peek at the 2025 340B Spending Numbers
The Obesity-News Deluge Continues With a Deeper Look at Cash-Pay Realities and Two Interviews With the Novo CEO
How HRSA Is Thinking About Rebate Models, Take Two
Why the End of the CIA’s World Factbook Is a Problem for CMS’ Proposed MFN Pilots
Want Apex? This is the link to upgrade.
INFLECTION POINT (1)/ Smaller Companies Want to Strike MFN Agreements, but the White House Hasn’t Established a Pathway for Dealmaking
Last year, 17 pharma companies signed deals with the White House in which companies provided some concessions -- mostly around Medicaid prices and the launch price of future meds -- in return for freedom from tariffs and future administration efforts to control prices.
It’s generally been seen as a pretty good deal for the companies involved, offering a sense of certainty if nothing else.
But there are a lot more than 17 pharmaceutical companies, and one of the big questions of the past two months is what happens to everyone else. Reuters explored that topic in a piece yesterday.
The takeaway isn’t that smaller biopharma companies want access to the same kinds of deals that the big firms got. We already know that. What’s notable in the story is the general lack of interest being shown by the government.
White House provided a quote -- “The Trump administration of course wants to negotiate deals that meaningfully lower drug prices for American patients with every pharmaceutical company” -- but industry hasn’t received any guidance on how to make those talks happen.
The lack of a roadmap appears to be a driving force behind the creation of a new membership organization for 10 smaller pharma companies, the Midsized Biotech Alliance of America.
There is general optimism that something will be worked out. The Reuters piece quotes a Bayer executive who said it would be “very odd” not to treat all manufacturers the same.
Still … we’re living in a very odd time.
INFLECTION POINT (2)/ Nearly All Oncologists Have Big Problems With Step Therapy, New Survey Finds
It’s not news that utilization management -- think prior authorization and step therapy -- is unpopular. Last week, there was KFF data showing that, next to cost, prior authorizations were the single biggest burden felt by patients, and that nearly half of all patients had care disrupted by prior authorizations.
Doctors aren’t any happier. A new Community Oncology Alliance poll found that oncologists don’t think that step therapy policies are aligned with clinical guidelines, that patients are often harmed, and that offices have had to add staff to keep up with the paperwork.
The significance of the COA poll isn’t the findings themselves. It’s the near-unanimity of the respondents. Are step therapy rules backed by clinical data? Ninety-one percent said no. Ever seen a patient struggle due to step therapy? More than 96% said yes. Need more employees? Absolutely, according to 89% of those polled.
Every datapoint seems to be pointing toward the broad conclusion that utilization management has gone too far. The only question is whether insurance companies will begin to back off or whether Congress -- where this is a hot-button issue -- will make them.
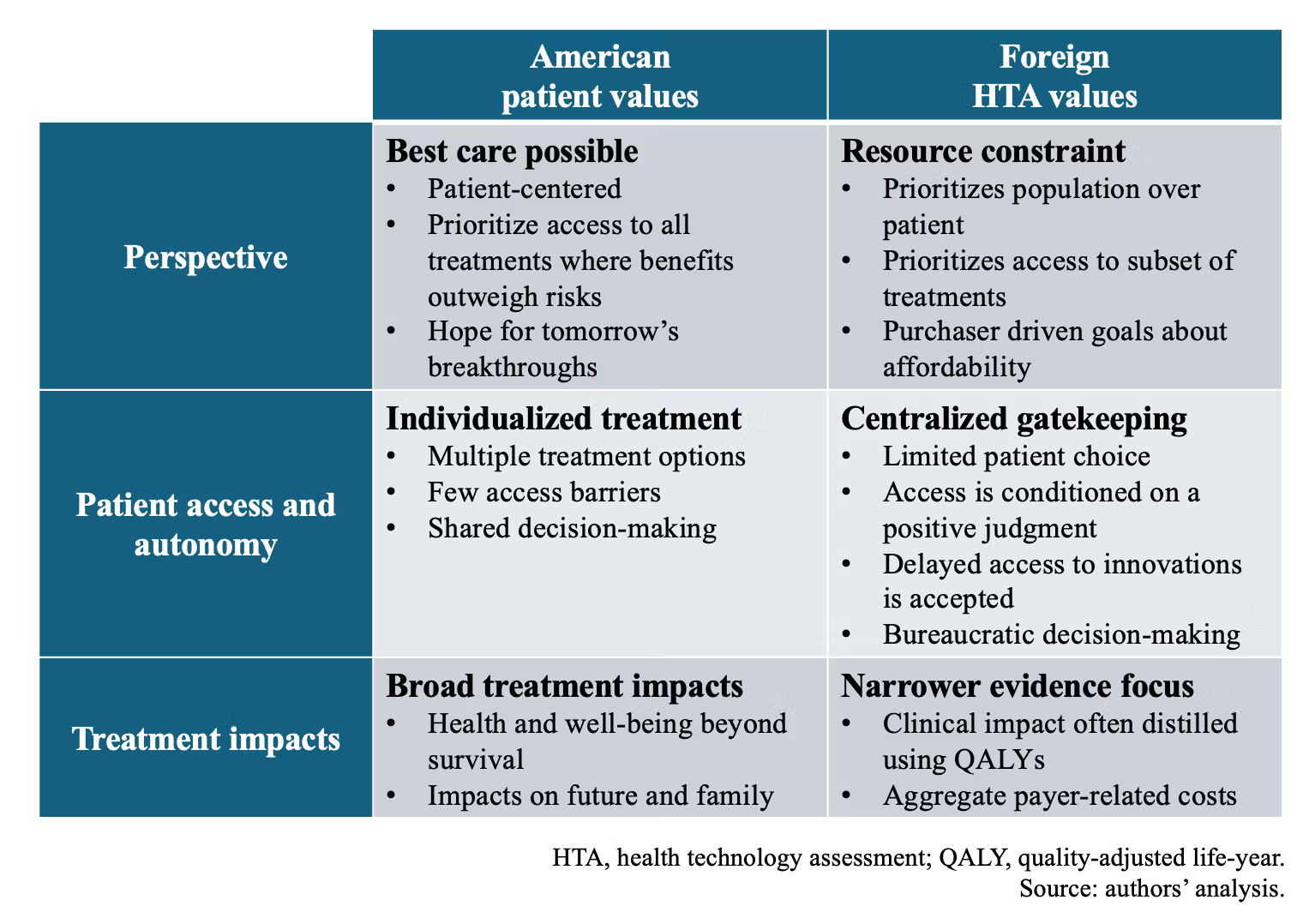
QUICK TAKES/ Quantifying How Americans Value Meds Differently Than Europeans, a New IRA Lawsuit, and a Modest Proposal for Talking About Drug Prices
I frequently make the point around here that MFN policies explicitly outsource decisions around drug pricing to countries that have fundamentally different economies and fundamentally different values.
But saying American “values” are different feels a little vaporous. How, exactly, do those in the United States view health care’s costs and benefits differently?
There is now an elegant answer to that question, via a new publication in the American Journal of Managed Care by a team from the National Pharmaceutical Council. The whole thing is worth a read, but for those who are caffeinated and must rush, this chart distills things nicely.
ELSEWHERE:
Health policy experts don’t think, by and large, that the administration’s MFN approach -- even if implemented -- would actually work. That’s the conclusion from a Cornell Health Policy Center survey of this all-star group of wonks. Only 50% thought that net prices would even go down, and a large majority didn’t think access would improve for anyone.
I’m a fan of the argument, from Utah’s Joey Mattingly, that we need some sort of standard for the way we talk about drug prices. In a world with list prices, net prices, cash-pay prices, out-of-pocket prices, and more, defining terms has never been more important.
“At a minimum,” Mattingly wrote, “any public discussion of a drug’s price should clearly specify which price is being reported along with who the price applies to, including relevant eligibility criteria and access pathways.AbbVie has filed suit against the government, objecting to CMS’ selection of Botox for the next round of Medicare “negotiations.” CMS is barred from imposing price controls on “plasma-derived products,” and AbbVie argued that one of the three ingredients in Botox is plasma-derived human serum albumin.
Human albumin is clearly listed in the Botox label as a component of the drug, albeit as an inactive ingredient. I expect pages and pages of arguments over the next few months about whether the “plasma-derived products” exemption covers only situations where human albumin is an active ingredient.
The odd-couple Senate pairing of Liz Warren and Josh Hawley is once again trying to ban companies from owning both an insurance company and a PBM. Their new bill would also restrict the ability of drug wholesalers to own provider groups.
There is an increasingly deep literature on the victims of 340B. We know that employers take a hit from lost rebates. Ditto many state Medicaid programs. And now, via this IQVIA report, the impact on state employee health plans has been quantified. The overall number -- $1 billion in additional spend due to 340B overcharges -- should be attention-grabbing, but the real benefit of the analysis might be the state-by-state breakdown.
Cost Curve is produced by Reid Strategic, a consultancy that helps companies and organizations in life sciences communicate more clearly and more loudly about issues of value, access, and pricing. We offer a range of services, from strategic planning to tactical execution, designed to shatter the complexity that hampers constructive conversations.
To learn more about how Reid Strategic can help you, email Brian Reid at [email protected].


